Si 100
hamdail plc
What is Selenite?
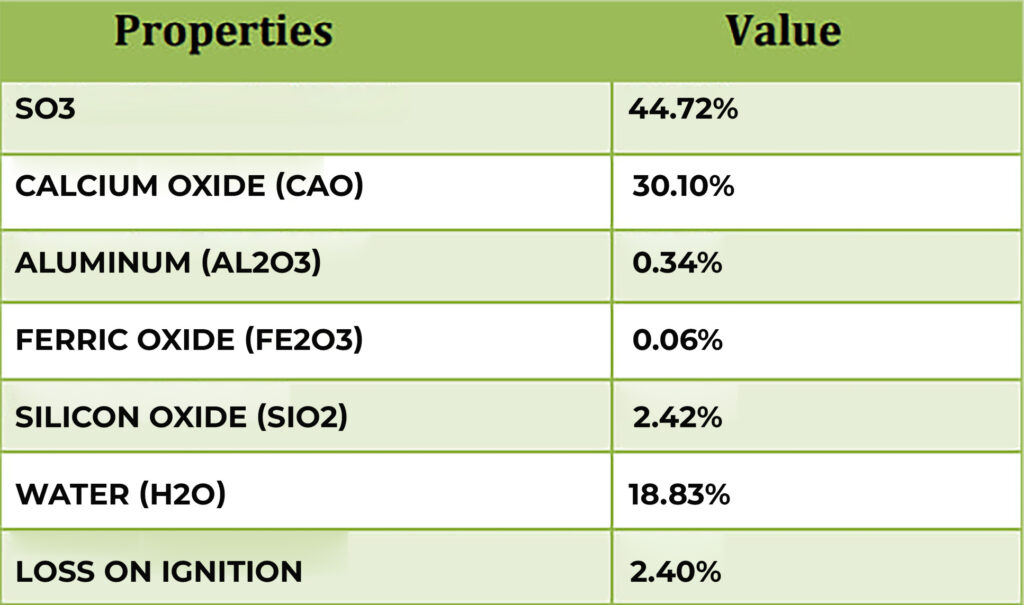
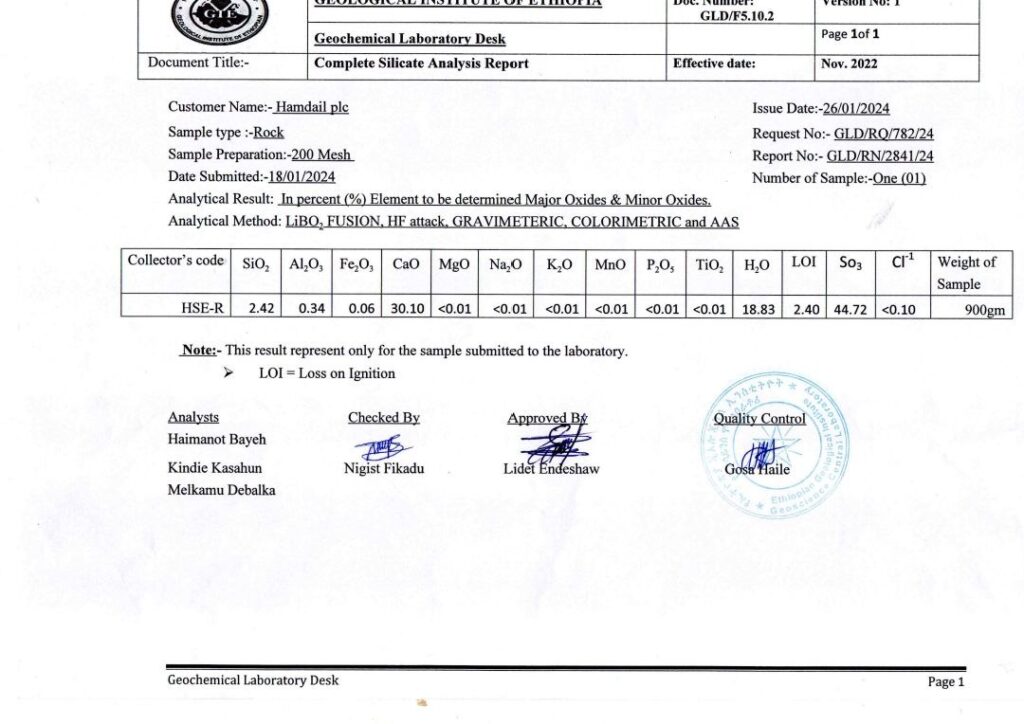
Selenite is a crystallized form of hydrous calcium sulfate (CaSO4.2H2O); it is a translucent variety of gypsum prized for its pearly luster and versatile applications ranging from high-end decor and holistic wellness to industrial use.

Types
Our Ultra white calcium carbonate is a premium quality product characterized by its exceptional whiteness and high purity. It is sourced from the finest natural quarries and processed using state-of-the-art technology to produce a product that meets the most stringent quality standards.
Our Off-white calcium carbonate is a high-quality mineral compound widely used in various industrial applications. It is sourced from natural deposits and processed using advanced technologies to achieve a consistent and uniform particle size distribution